Lawyers for the patients also wrote in the suit that Quest has “paid kick-backs to medical providers in the form of dramatically below-cost billings,” schemed with “two major private health insurers to suppress its competition,” and “acquired its competitors for plan/outpatient billing in order to eliminate their competition.”įounded in 1967 as Metropolitan Pathology Laboratory in New York, the modern Quest became a public company in 1997 in a spin-off from the Corning conglomerate. Lawyers for Eastman, Cruz and Mendez argue that Quest billed them at unfair, above-market prices that their health plans passed onto them through increased outpatient payment obligations. Three patients in Northern California are suing Quest Diagnostics, claiming the company has taken over most outpatient diagnostic services in the region by acquiring would-be rivals, inducing doctor referrals and colluding with payers like Aetna and Blue Shield of California to stifle competition.Ĭolleen Eastman, Christi Cruz and Carmen Mendez, filed the federal antitrust lawsuit and class action complaint against Quest in U.S. 
This chart shows different kinds of adulterants, ways a urine specimen can be diluted or substituted and products used by donors in attempts to “cleanse” their specimen prior to a urine drug test.Laboratory giant Quest Diagnostics is facing allegations that it unfairly worked with payers and physicians to create a monopoly in one the nation’s largest markets. All “Invalid” specimens,” based on analytical test results, are automatically tested twice on a different portion of the urine specimen to ensure accuracy. We monitor and adjust our testing as necessary when new adulterants become commonplace. The use of oxidizing adulterants – such as nitrites, chromates and halogens (e.g., bleach and iodine) – is one of the most common ways donors try to “cheat a drug test.” Quest Diagnostics is one of the few laboratories to offer oxidant identification for some of the most common oxidizing adulterants and is available when requested by the customer or Medical Review Officer. Substituted specimen: A urine specimen with creatinine and specific gravity values that are so diminished or so divergent that they are not consistent with normal human urine. Normal values for the specific gravity of human urine range from approximately 1.0020 to approximately 1.0200. For urine, the specific gravity is a measure of the concentration of dissolved particles in the urine. Specific gravity is a measure of the density of a substance compared to the density of water. HHS set the program cutoffs for pH based on a physiological range of approximately 4.5 to 9. Human urine is usually near neutral (pH 7), although some biomedical conditions affect urine pH. 
Examples of these agents include, but are not limited to, nitrites, pyridinium chlorochromate, chromium (VI), bleach, iodine, halogens, peroxidase and peroxide. Oxidizing adulterant: A substance that acts alone or in combination with other substances to oxidize drugs or drug metabolites to prevent the detection of the drugs or drug metabolites, or affects the reagents in either the initial or confirmatory drug test. 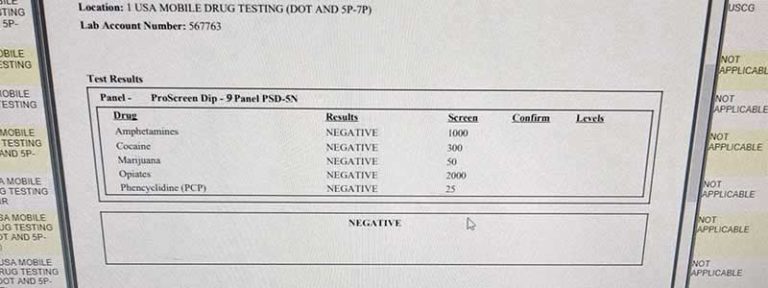
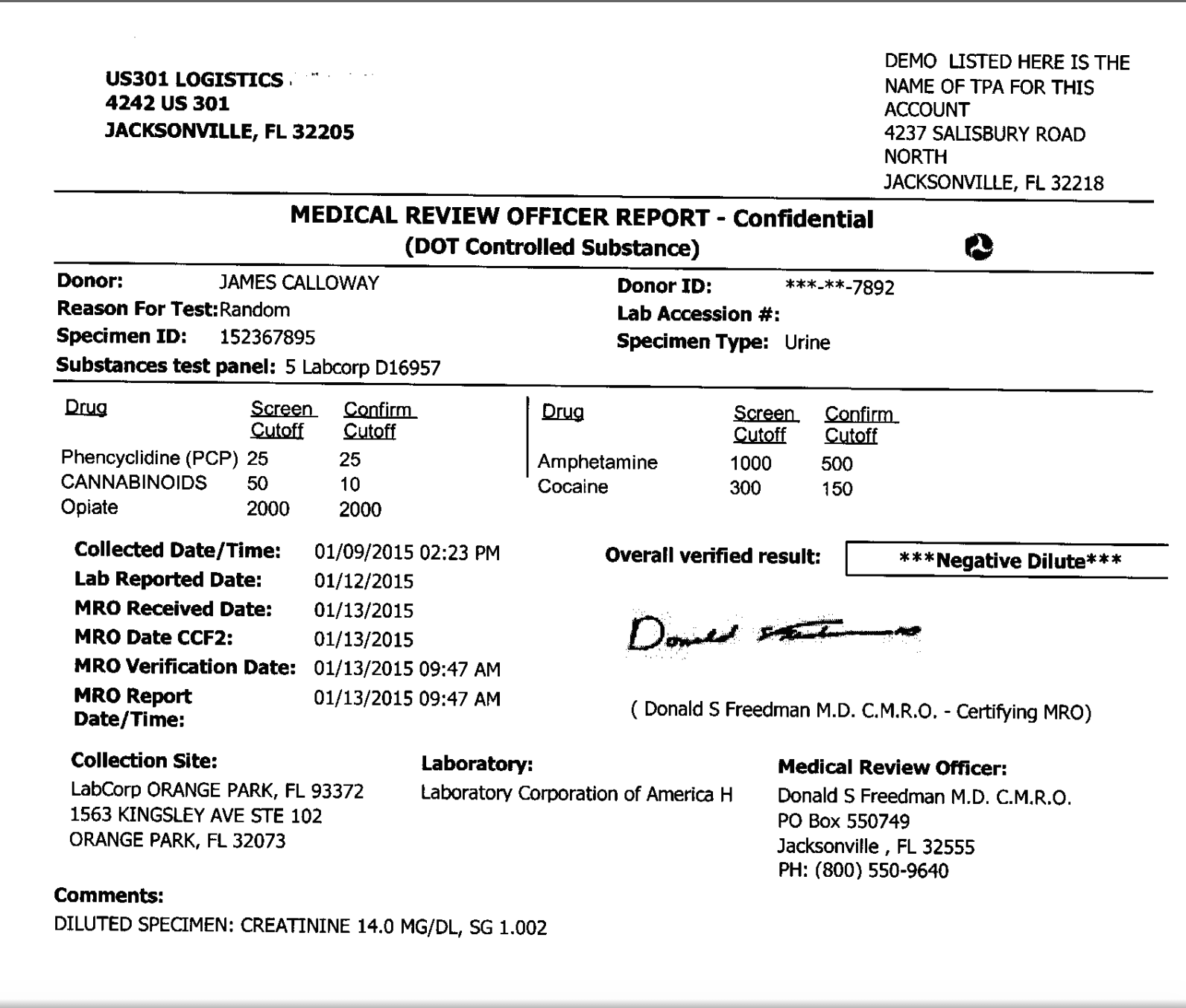
Invalid result: Refers to the result reported by a laboratory for a urine specimen that contains an unidentified adulterant, contains an unidentified interfering substance, has an abnormal physical characteristic, or has an endogenous substance at an abnormal concentration that prevents the laboratory from completing testing or obtaining a valid drug test result. Normal human urine creatinine concentrations are at or above 20 mg/dL.ĭilute specimen: A urine specimen with creatinine and specific gravity values that are lower than expected for human urine. Here are definitions to provide a better understanding of terms related to specimen validity testing:Īdulterated specimen: A urine specimen containing a substance that is not a normal constituent or containing an endogenous substance at a concentration that is not a normal physiological concentration.Ĭreatinine is endogenously produced and cleared from the body by the kidneys. The US Department of Health and Human Services (HHS) Substance Abuse and Mental Health Services Administration (SAMHSA) defines drug testing terminology in its Mandatory Guidelines for Federal Workplace Drug Testing Programs and the Medical Review Officer Manual for Federal Agency Workplace Drug Testing Programs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
